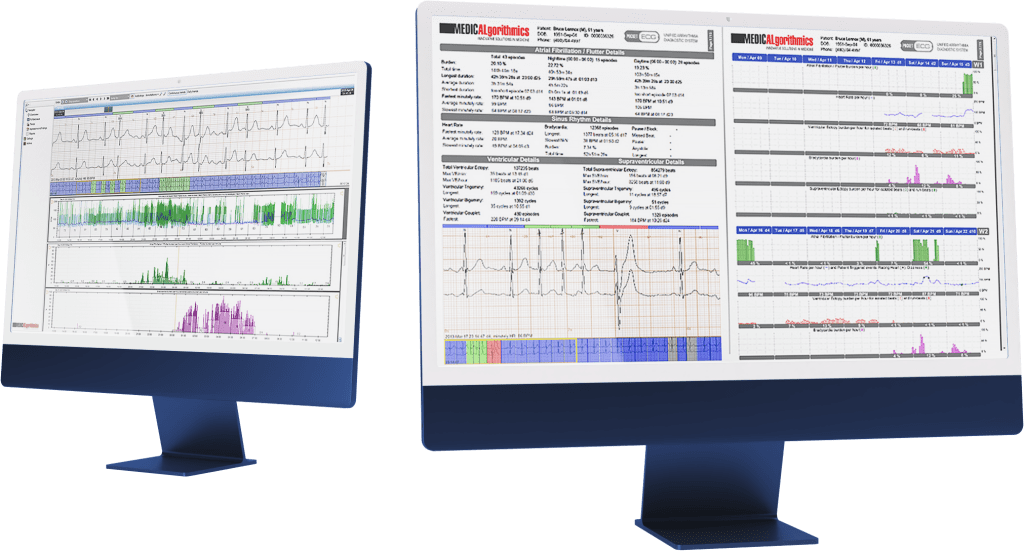
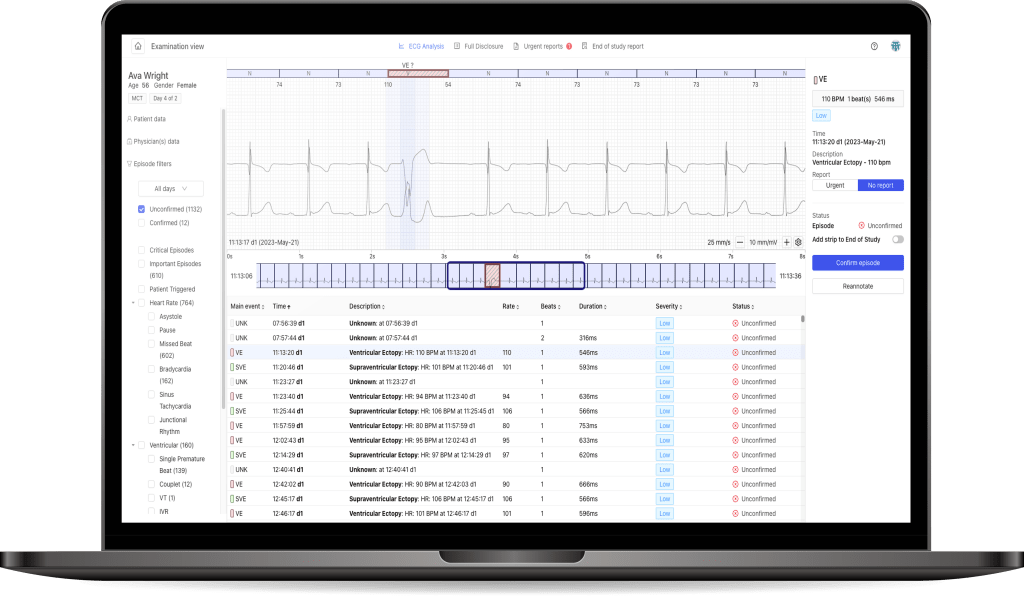
Medicalgorithmics, a cardiac medtech listed on the sWIG80 index, has achieved another milestone by initiating the CE certification process for its VCAST technology, designed for non-invasive diagnosis of coronary artery disease on coronary CT scans. The AI-based VCAST technology is being developed by Kardiolytics Inc., a subsidiary of Medicalgorithmics.
Medicalgorithmics has reached an important milestone in the development of the VCAST technology, as outlined in its new development strategy announced this past June. On October 3rd, by signing an agreement with the certification body TÜV Rheinland Polska, the medtech company formally began the compliance assessment process of the technology with the EU medical service requirements. Successful completion of this assessment will culminate in the granting of a CE certificate, essential for the commercial launch of VCAST in the European Union market.
“The start of the certification process is a significant milestone, bringing us closer to commercializing VCAST technology within the European Union. Upon obtaining the CE certificate, which confirms VCAST’s compliance with EU requirements for medical technology, we will register the system with the Office for Registration of Medicinal Products, Medical Devices, and Biocidal Products, enabling us to offer VCAST technology in Poland. Additionally, after obtaining the CE, we will apply to regulators in other EU countries, as well as in Australia, the UK, and Switzerland, which we consider key in terms of sales potential. This ensures that by 2024, VCAST will be fully available to partners in these countries,” says Przemysław Tadla, Board Member in charge of technology at Medicalgorithmics and COO at Kardiolytics.
This past September, Medicalgorithmics signed a letter of intent with the Diagnostyka Group to utilize VCAST technology for diagnosing coronary disease at the partner’s diagnostic facilities.
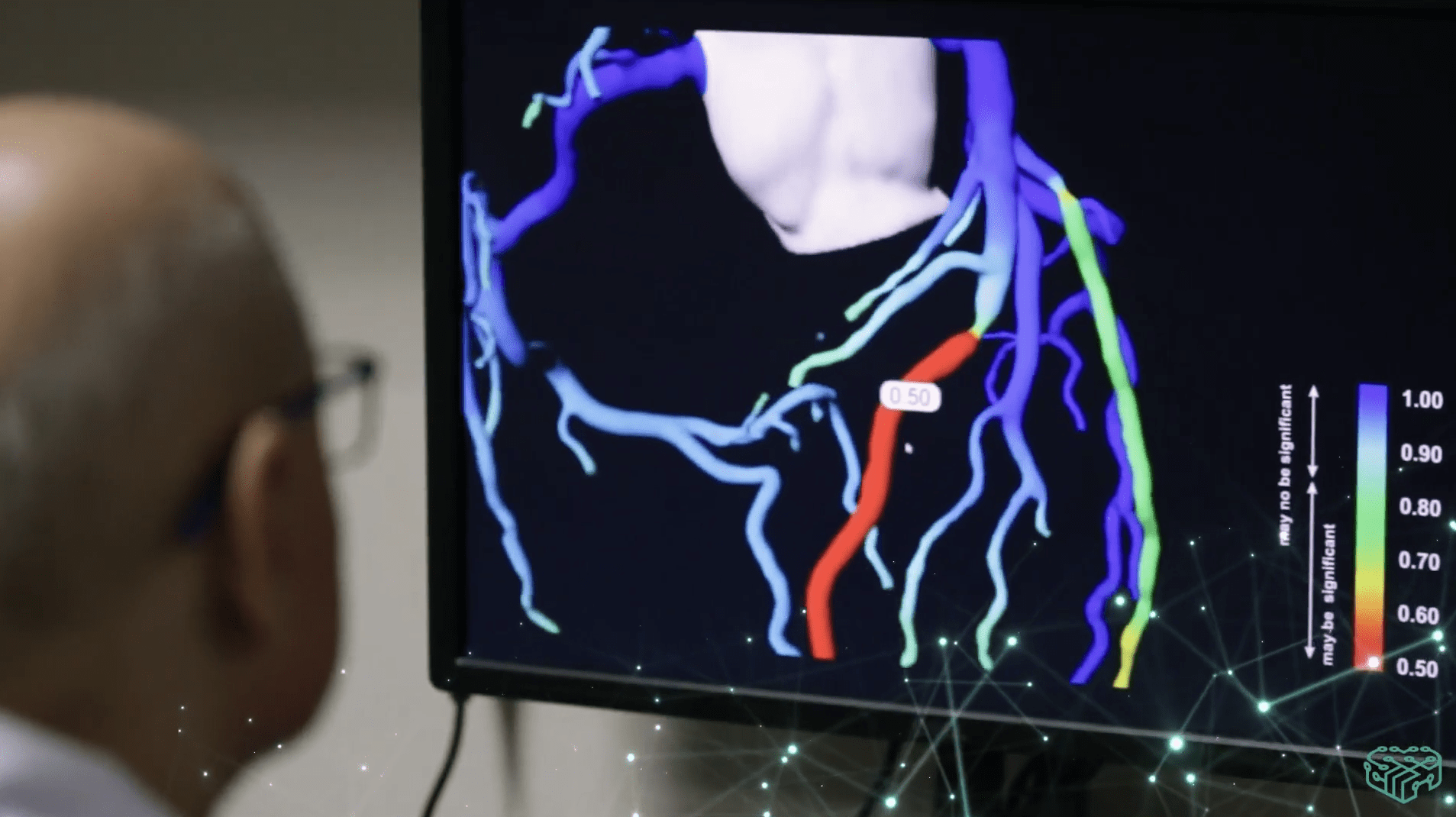
The Virtual Stress Test of the Heart (VCAST), a patented system developed by Kardiolytics—a subsidiary of Medicalgorithmics—facilitates non-invasive analysis of coronary vessels for diagnosing coronary heart disease. Diagnoses are based on heart computer tomography data, analyzed by a suite of AI algorithms developed by Kardiolytics. This allows physicians to quickly assess a patient’s coronary artery condition without consistently resorting to the risky, invasive, and costly coronary angiography procedure.
“The globally increasing incidence of coronary disease and an aging patient population in developed countries underscore the immense market potential we see for VCAST technology. Just in the US, it may be used in diagnosing and treating over 10 million cardiological patients. Thus, I’m excited that after the developmental phase, Medicalgorithmics and Kardiolytics have taken the first step towards commercializing the VCAST system. The technology of personalized, three-dimensional modeling of coronary arteries using our proprietary AI system is set to become another significant business avenue for Medicalgorithmics alongside arrhythmia diagnosis,” says Kris Siemionow, a member of Medicalgorithmics’ supervisory board and managing partner at Biofund Capital Management LLC, the company’s main shareholder.
Kardiolytics, a company developing AI-based medical solutions, joined the Medicalgorithmics capital group in November 2022. This was part of the Biofund Capital Management LLC’s investment in the cardiac medtech.
In line with its development strategy announced this past June, Medicalgorithmics plans to submit to the US Food and Drug Administration (FDA) for VCAST technology’s FDA registration and entry into the US market in the first half of 2024.
***End***
For more information, please contact:
Media:
Mariusz Gawrychowski
InnerValue
+48 501 520 598
Investors:
Robert Mrozowski
InnerValue
+48 513 083 322